You wouldn't normally write them any differently from the other orbitals. 
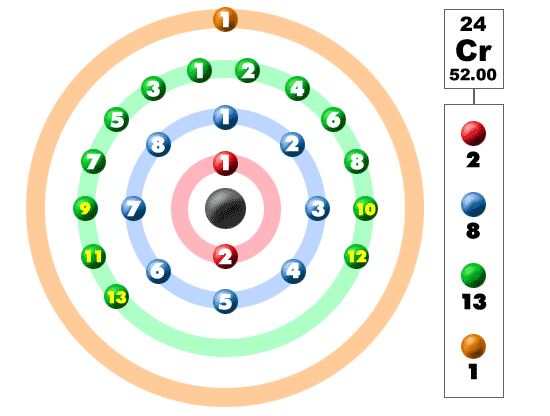
Note: The orbitals where something new is happening are shown in bold type. These levels all have the same energy, and so the electrons go in singly at first. Beryllium adds a second electron to this same level - 1s 2 2s 2. Lithium has an electronic structure of 1s 2 2s 1. Lithium's electron goes into the 2s orbital because that has a lower energy than the 2p orbitals. Now we need to start filling the second level, and hence start the second period. Hydrogen has its only electron in the 1s orbital - 1s 1, and at helium the first level is completely full - 1s 2. If you are studying a UK-based syllabus and haven't got them, follow this link to find out how to get hold of them. Important! You must have a copy of your syllabus and copies of recent exam papers. This page looks in detail at the elements in the shortened version of the Periodic Table above, and then shows how you could work out the structures of some bigger atoms. It is important that you look through past exam papers as well as your syllabus so that you can judge how hard the questions are likely to get. After barium you have to worry about f orbitals as well as s, p and d orbitals - and that's a problem for chemistry at a higher level. UK syllabuses for 16 - 18 year olds tend to stop at krypton when it comes to writing electronic structures, but it is possible that you could be asked for structures for elements up as far as barium. This isn't a mistake, but an effect of converting my original diagram into a lower quality gif image for efficient web use. Note: On some screens the V for vanadium (element 23) may look a bit like a Y. Relating orbital filling to the Periodic Table Important! If you haven't already read the page on atomic orbitals you should follow this link before you go any further. If you want to look at the electronic structures of simple monatomic ions (such as Cl -, Ca 2+ and Cr 3+), you will find a link at the bottom of the page. It assumes that you know about simple atomic orbitals - at least as far as the way they are named, and their relative energies. Wild electron configurations also mean you have a good chance at a variety of valence numbers.This page explores how you write electronic structures for atoms using s, p, and d notation. Potassium, chromium, and copper are the only elements in the fourth period that have one electron in the outermost shell. While vanadium (to the left) added one electron to its third shell, chromium adds its electron to the third shell and has one electron from the fourth shell bumped down. Being a transition metal, the electron configuration is unique. The fourth row of the periodic table has transition metals ranging from scandium (21) to zinc (30).Ĭhromium is number twenty-four on the periodic table and located in the same column with molybdenum and tungsten. Remember that the first eight were placed during our trip through the third period/row. For the fourth period/row, all of these electrons build the third shell to a maximum of 18 electrons. This element is one of the transition elements that doesn't place the additional electrons in the outer shell, but in the one underneath. So remember when you look at our breakdown that the electrons aren't always in a nice neat order as shown here. They are found in clouds that can have different shapes that include spheres and dumbbell-like shapes. As you learn more about atomic structure, you will learn that the electrons don't stay in defined areas around the nucleus. The electrons like to be in separate shells/orbitals. In an atom, the electrons spin around the center, also called the nucleus. Each of those colored balls is an electron. If you think this is a little over your head, go back and look at the elements 1-18 that have organizations that are a little more simple. Let's take a look at the arrangements of electrons in the basic elements (left and right sides of the table) of period four and the more complex arrangements of the transition elements (in the middle of the row). You may have an easy way to know the number of electrons in a neutral atom, but the placement of those electrons gets a little more complex. 
Now we're working with the fourth period/row in the table of elements. It tells you the mass of one atom, how many pieces are inside, and where it should be placed on the periodic table. That box on the left has all of the information you need to know about one element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
